Diagnostic-imaging refers to technology-driven medical practice through which the health situation of a patient is investigated by healthcare personnel. Imaging devices are utilized for capturing images through x-raying, scanning and ultra-sounding of internal organs for onward medical prediction, interpretation or diagnostics. Due to the changing complexity of diseases, medical imaging has transformed through the years to meet up with the transitional demands of emergent and occurring modalities in a format that is structural. Through the sophistication of imaging technology, it is possible to transfer patients’ demographics, examination-properties, imageries, or numerical-data in secured and highly efficient ways. This study is concerned with predicting treatment response using medical imaging information. It emerged from this study that predicting tumor response to medication is a complex exercise that cannot rely on baseline image information alone hence the need to consider a combination of imaging biomarkers.
Method and methodology
This study was quantitative in nature. An opportunistic sample of ten patients with 26 colorectal liver metastases was used as the population of study. They each had double DCE-MRI examinations before the commencement of 1st line medication with bevacizumab and FOLFOX6. The study used non-linear regression model during data analysis. All the biomarkers used to evaluate the size of the tumor and the blood supply of the growth were identified and a regression was conducted against the post-treatment change in the volume of the tumor with therapy being repeated five times over. This was in order for us to come up with a non-linear model for defining the observed change in comparison with imaging indicators of measurement. The resulting model’s predictive function was analyzed by using the leave-one-out tests.
Discussion
Data that is collected from quantitative imaging studies can be utilized in the prediction of how a tumour will respond to therapy. This information indicates that the blood-supply structure and distribution have a very important function in the analysis of tumor response to treatment.
Key words: Diagnostic-Imaging; Imaging Information; Medical Prediction; Treatment Response
Predicting Treatment Response Using Medical Imaging Information
Introduction
Radiological studies have for years have been concerned with the imaging and estimation of blood flow as well as in establishing the characteristic structure of veins and capillaries within certain tissues (Meyer et. al., 2009). Earlier experiments revealed that dynamic contrast imaging conducted using basic radiography techniques was a greatly powerful method for use in identifying and characterizing various tissues (George et. al., 2001: Meyer et. al., 2009). Much later, and with advancing technology, axial imaging tools were developed and this provided even more methods for the diagnosis and classification of tumers (Link et al, 2001). These tools were adopted for use in oncology units especially because the study of blood supply patterns involved in tissue calcification had become of biological relevance. Over the past two decades, intensive studies have been conducted on the aspect of using imaging techniques to diagnose and classify lesions depending on the development of new blood vessels from already existing venous systems (Lode et al, 1998). This process of vasculature development is known as angiogenesis. One of the greatest observations recorded over this period is that imaging techniques can reliably be used in the provision of prognostic data that is different from basic histological grading strategies in various tumors. Of even greater importance is the development of mechanisms that can be used to control the development of new vasculature. The consequence of such a discovery was that there was an even bigger need for surrogate markers of angiogenesis which could also be effectively used in the development of medications (Link et al, 2001). The ability of MRI to generate images without the usage of radiation, using various contrast techniques was already giving the technology even more clinical acclaim. The development of methods for easy acquisition of images provided opportunities for the assessment of time changes in tissue vasculature (Lode et al, 1998). Because of the increase in demand and the development of technology for specialized assessments, angiogenic imaging went through a vigorous growth spurt, of which Dynamic Contrast-Enhanced MRI (DCE-MRI) is the most popular. DCE-MRI has its foundation in the development of drugs as well as basic oncology research. It should, however, be noted that other imaging techniques (which will be briefly mentioned in this study) have emerged and are providing vigorous competition for DCE-MRI.
The aims of the project
Once a VEGF inhibitor has been provided to deal with a cancerous tumor, the reduction in the size of the swelling can be pre-determined using measurements that are picked up from MRI scans. The regression model was used to analyze the issues listed below:
- To describe the reduction in the size of the tumor as a non-linear model of taking measurement using imaging techniques.
- In general, measurements may be impacted negatively because of error (for instance the movement of the patient while in the scanning machine). This project thus aims to use regression and measurement in tandem with each other.
- Challenges tend to arise in imaging analyses and may present in the form of missing data. These setbacks were factored into the regression strategy.
- A comparison was made between linear and non-linear models of prediction.
Approach
This study takes the approach of a retrospective study that reviews patient records to find the critical success factors of imaging biomarkers. This was achieved through embarking on an evidence-based investigation of both major components in the treatment prediction regimen. At the end of the project, one is in a position to present an analysis and offer rigorous recommendations based on evidence-based inquiry techniques. The advantages of this approach include the reliability and consistency of the data since these have to be recorded under the guidance of center staff at the time of admission. This is information that patients provided in the forms which patients complete on admission.
Relevance of Project to the Practice
The project will serve to advance scholarly findings in the field of imaging technologies and their effectiveness. This will be presented principally by showing how much of the prediction-to-treatment success rate can be attributed to the kind of imaging technique used. The effectiveness of the combination of various program components shall also be evaluated. This is a significant element since much of the available literature is based on investigations on single factor pairs, i.e., the success criterion and one influential variable. This strategy will in effect go a long way in enhancing evidence based engineering.
Project evaluation
Rationale for and Importance of Evaluation
Accurate prediction of treatment outcome is an essential part in the decision making process of cancer treatment. When treatment outcomes are accurately predicted, health care professionals are able to make necessary treatment changes that are more beneficial to patients. However, accurate prediction can only be possible if aided by appropriate facilities and technologies. For the sake of their patients and staff, imaging system center administrators need to regularly evaluate services and systems with evidence-based feedback in order to establish what works and what needs to be replaced.
The UK’s National Institute for Clinical Excellence (NICE) regulates health services by providing guidance on how to promote good health and prevent/ treat ill health. Despite its role in ensuring good health, NICE recently became under criticism for failing to approve bevacizumab (Avastin); a drug that is believed to have recommendable cost effective benefits in the treatment of breast cancer. This, therefore calls for studies to accurately predict treatment outcomes of cancer patients to be able to decide on what works best.
Objectives
In general, this program evaluation is prognostic and serves as a benchmark. The evaluator seeks to analyze which imaging systems are more often associated with accurate prediction of treatment response. This is particularly important, given that a majority of the patients in most oncology facilities need extreme accuracy in the prediction, diagnosis, and administration of treatment regimes for decent success rates to be recorded.
The treatment modalities and outcomes patients experience in imaging approaches are beyond the control of the evaluator. Nonetheless, the penultimate stage of this project will include post-hoc information gathering about those techniques tagged with the best and worst success rates.
Prospective Benefits
Knowledge of the extent of a tumor is fundamental in the staging of the solid tumor and as such important in the selection of treatment methods as well as the assessment of the applicability of resection in addition to the prediction of clinical results. The concurrent usage of various pre-treatment measures obtained from DCE-MRI parameter maps may be used reliably in the predication of tumor shrinkage. This is in response to the clinical low-error combination of bevacizumab and cytotoxic chemotherapy. All in all effort must be made to try and implement the recommendations made in this benchmarking program evaluation project. This can bring about major benefits, top on the list being: encouraging a thorough review of treatment program prediction components of various modalities.
The Benchmark Criteria: Short- and Long-run Effectiveness
Patient progress towards being free of cancer is, of course, the prime consideration in outcome-based evaluation. The short-term effectiveness of this project is to identify effective prediction methods that can lead to correct diagnosis and treatment of cancer. After stabilization and recovery, the patient should be able to enter therapy in a cancer-free state. Cancer-free status may be self-reported and supported by physicians providing pain management and general medical care, as well as by responsible personnel at treatment centers being attended.
The study is presented in four sections as illustrated below:
- An introduction chapter which contain basic information about the project
- A background chapter which contain all necessary researched information to set the foundation for the study
- A methodology section that will contain all the experiments and tests that were conducted in sourcing of data for the study
- A Conclusions section, which will present the conclusions drawn from the findings obtained from the study
Background
Background
Much of the credit to medical imaging predictions addresses cancerous diseases. Studies have shown that at least twenty-five percent of patients with a tumor that is bigger than three centimeters have benefited from neoadjunctive chemotherapy which is highly dependent on predictions using medical imaging (Buchegger et. al, 2000). It is further noted from studies that over one quarter of patients suffering from cancer do not show any positive response to chemotherapy (Buchegger et. al, 2000). As such it is of paramount importance to use prediction techniques to identify such patients (Landheer et. al., 2001). A tool that aids in predictions using medical imaging has been noted to include techniques that are used in detecting how a cancerous growth responds to chemotherapy attained through the usage of Positron Emission Tomography (PET) scans (Geoghegan and Scheele, 1999).
The reliability of medical imaging is influenced by artificial intelligence through articulate structuring of models based on earlier investigations of a particular circumstance in algorithm formats that could be fed with finite collections of observations (Landheer et. al., 2001). This dissertation will present studies on predicting treatment response using medical imaging information with specified inclination to algorithms of data acquired through clear data analyses. In their research, Glover et al. (2000) studied an optimal time for medical-imaging utilizing the PET device for prediction of patients’ response to neoadjunctive-chemotherapy. The study was effective in its measurement of mean standards for up-taking midpoint-neoadjunctive values in chemotherapy which reflected an approximately seventy-seven percent low-patients response, and a hundred percent high response which achieved a 93% area ROC. The dissertation will adopt similar analytical approaches in its presentation of the subject mater.
Introduction to Cancer, Colorectal Cancer & Liver Metastases
Studies have reviewed that the presence of metastatic-disease which is found within the liver is recurrent in oncology (Primrose, 2002). Epithelial cancer finds the liver a very good site for growth where it spreads metastatic-natured, only seconding the lymph-nodal region (Primrose, 2002). In any case, research has not proven adequately the prevalence of the disease. For instance, Curley et. al., augurs in line with this saying that approximately 20-25% of patients with colorectal cancer show signs of liver metastases during the diagnosis phase (Curley et. al., 1999).
This complexity has made the adaptation of medical imaging vital in predicting the development of colorectal cancer. When the prediction turns positive and there is an early diagnosis of the liver-metastases, this could go a long way to be of great help to a cancerous patient. Bloom et al. (1999) noted that liver metastases are an indication that the primary tumor cannot be resected.
Considering patients with this condition, using the method of chemotherapy has always been a preferred option. Only with a certain number of malignancies, re-sectioning the liver’s metastases would then indicate an improvement of chances for the patient’s survival (Johns and Houlston, 2001). Studies by Zealley et. al (2001) have stressed that the presence of liver metastases for patients with colorectal cancer indicate a need for surgical resection.
The prediction of treatment on a patient at this instance would depend on the localization/number of the liver-metastases and there would be a consideration of the several clinical-parameters that relates to the patient. The chances for resectioning colorectal-patients with liver-metastases stand at 30% (Heslin et. al., 2001). In any case, the possibility for the patients to survive in five years is just about 30% to 48% as compared to the five-percent of chances for survival for patients with liver-metastases which is not liver-surgery amenable (Popov et. al., 2002). It is the responsibility of radiologic-imaging to analyze the condition of the liver by assessing the availability or the non-availability of liver-metastases when considering surgery of better predictions as well as for evaluating successes of other patients’ chemotherapy. Even though there has been the use of transabdominal-sonography in assessing the liver, the method is not without its shortcomings: there has to be a consideration of the operator’s level of understanding which in most cases reflects the reviewer of equivocal-results in patients who (induced with chemotherapy) express fatty liver infiltrations.
Biomarkers of response (Imaging Methods)
Medical-imaging is carried out using x-ray, ultra-sound, magnetic-resonance as well as radionuclide. Each of these methods has its own strengths; for example, through displaying anatomical structures in a way that would show the physiological activities of the organ/tissues. These basic methods and a number of others are discussed below:
Magnetic Resonance Imaging (MRI)
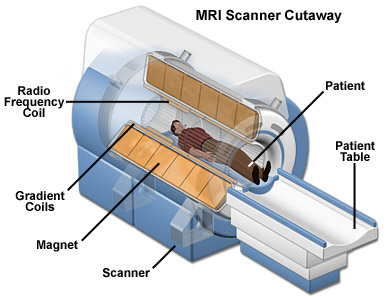
This is a technique that utilizes magnetic fields and radio wave to make graphical representations of organs and structures within the body. In a number of cases, MRI presents varying data from structures that are not visible with X-rays, ultrasounds or CT scans. MRI can be used to evaluate medical issues that cannot be viewed using other imaging techniques. MRI is important in the analysis of infections of the brain and spine. It also helps in studying the effects of stroke and the status of the spine after surgery. It is also important in the study of joints and muscular abnormalities.
The human body is to a large extent made up of water molecules. Therefore, when a patient is placed into the magnetic field of the MRI scanning area, the magnetic bearings of the spin of protons align themselves with the flow of the magnetic field (Figure 1). This change in flow generates a radio frequency that ends up producing a bigger electromagnetic field. The photons of the electromagnetic field have some energy which is known as resonance frequency which enables them to be sucked in and alter the rotation of to the protons that are aligned in the body. The power of the magnetic field determines the frequency of resonance for the protons. Once the RF energy is switched off, the protons that had taken in energy return back to the original low-energy state of rotation. A H2 dipole comprises double spins- one high and the other low. The dipole and magnetic domain are parallel to one another in low spin and vice-versa. The difference is released in form of energy as photons. The photons which are given out are picked up by the scanner in the same way that radio waves would. The relationship between the strength of the field and the rate of spin makes nuclear magnetic resonance ideal for imaging. Different tissues go back to equilibrium at varying rates and this allows the construction of images.
Dynamic Contrast-Enhanced MRI
This particular modality of perfusion-imaging makes use of repetition of imaging in tracking the emergence of contrasting agents that are diffused into body tissues within a period of time. According to studies by Choi et al this is achieved when: “…a paramagnetic contrast agent, gadolinium-DTPA, is injected intravenously circulates through the body and diffuses over time into the extravascular extracellular space” (2001). Consequently, as the average contrast agent volumes in a voxel goes up, so does the intensity from the same voxel (Choi et. al., 2001).
Through the knowledge of the characteristics of sequences of imaging, there is the possibility of converting relative-signal-increases into a quantifiable or measured contrasting-agent after a period of time. Curves can then be made use of in obtaining semi-quantitative analogs that would clearly identify the path of flow of blood and can be appropriately used for predicting the rate or level at which the patient is responding to treatment (Lorenz et. al., 1998). Equally, in a situation whereby a related concentration-curve is realized from arterial-blood, collective data could be used in realizing a better quantified index of the flow of blood as well as have a comprehensive knowledge of capillary permeably.
The utilization of exogenous extracellular material for establishing contrast has contributed to the great developments in the sensitivity of visibility of tumors using MRI. In addition, it has also been shown that the time-period of the contrast agent to build can help in the derivation of analytical data pertaining to the anatomy of tumor vasculature. This information is extremely important as it has been illustrated that the growth of a tumor is greatly dependent on its blood supply. Folkman (1990; 1995) also illustrated that the characteristics of tumor micro-venous and capillary supply is structurally different from that of other body tissues. As such, the control of blood supply to the tumor is regarded as one of the fundamental basics in the treatment of cancer. Most anti-cancer treatments work by either destroying all the veins and capillaries in a tumor or by impeding the development of new ones. Because the arresting of tumor growth is less likely to get rid of the growth, the procedures described above are more appropriately linked to the preliminary phase of treatment. When it comes to the assessment of short-term drug effects, DCE-MRI techniques, which are sensitive to the vasculature to the tumor are more appropriate in the quantification of features and characteristics that are of paramount importance.
MRI data acquisition
The patients were each scanned two times before treatment, as a means of assessing the reliability of imaging as an ideal method for treatment prediction. The focus of the scan was limited to the liver. The extended Tofts model (described by the equation below) was selected in the calculation of tumor vascularity, which in essence was the only usable method for prediction of treatment using MRI.
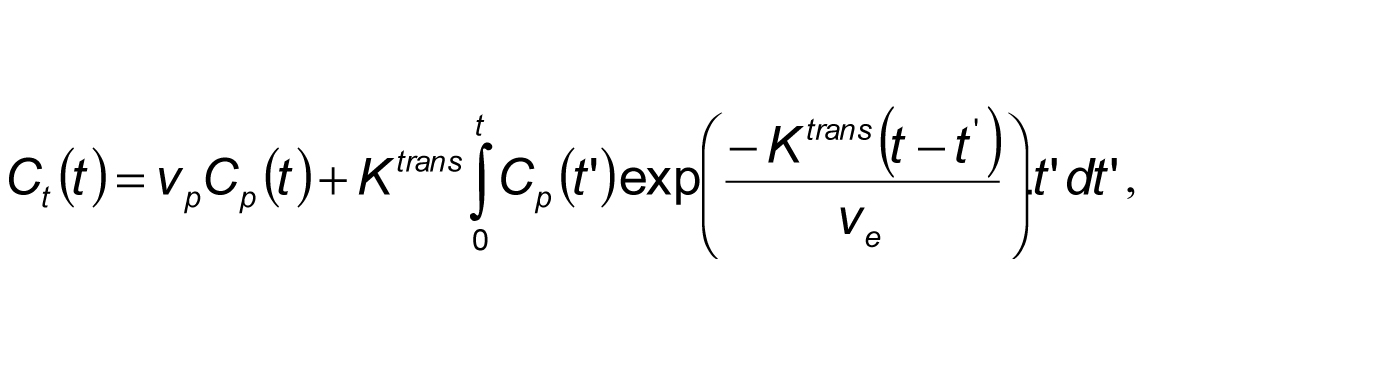
In this case Ct(t) was the amount of contrast agent at time t per voxel and Cp(t) was the amount of contrast agent contained in blood plasma of arteries. Ve represented the extravascular extracellur median fractional volume, Vp was the fractional plasma volume, and Ktrans represented the e median bulk transfer coefficient (O’Connor et al., 2011).
Other imaging methods
X-ray radiography
X-ray radiography uses a combination of gamma-rays and x-rays to generate a radiograph on a section of the body depicting changes in thickness, internal and external defects, and other fine details. This is the most popular technique of medical imaging. Images produced on film are used for the analysis of various body organs and structures.
Computerized Tomography (CT)
This is the highest technology for definition of medical-imaging which creates the cross-sectional view of an organ. It utilizes scanning technique undertake repeated imaging to obtain a complete image or a set of images of a section of the body then the technology is referred to as a scan (Ragnhammar et. al., 2001). Computerized Technology is used in the analysis of various body organs and tissues. The imaging method can also be used to produce 3D images of bones for orthopedic repair.
Multidetector-CT
Unlike MRI which utilizes magnetic fields and radio wave to make graphical representations of organs and structures within the body, MDCT utilizes scanning technology to obtain images of a section of the body. Presently with the introduction of Multidector-CT (MDCT) imaging-device, using CT for screening of patients’ lungs, liver, or lymph-node in oncology has become rather dramatic and on the increase. MR-imaging has continued to have defined point of reach through anatomic-coverage; even though the freshly introduced multi-channel. According to Fiorentini et al, coils that have an expansive coverage have made MDCT more competitive in regards to patient throughput (2000).
The multidetector-row-CT (MDCT) and CT have been very useful in imaging-modeling especially in regards to prediction of treatment. This is mainly because they are made up of a helical CT that has one detector row with a scanning speed of 0.8-1s per rotation (Neeleman et al., 2001).
The first MDCT was introduced in 1998 as a 4-row detector scanning system. The device has been very effective in covering the liver in ten to fourteen seconds, and feasibly reduces the possibility of motion-artifacts which could affect the scanning process while the patient is breathing. Presently, the forty to sixty-four row-MDCT scanning device has a 0.6mm detector device configured to it which presents a highly precise definition of the condition of the patient, and is of outmost value to predicting the response of the patient to treatment (Ruers et al., 2001). Equally, the device is fast and has enabled a reduced rotation-time to 0.33 seconds at most. This has been enabling liver scanning using submillimeter-collimation within the briefest two or three seconds of holding breath. Dependent on isotropic-voxels, reforming images through whatsoever plane is realizable without any loss of spatial resolution.
Fluoroscopy
This technique uses X-rays presented onto a cathode-ray tuve monitor so that everything under study is presented in detail. For example, the beating on the heart can be captured under this circumstance and analyzed.
Ultrasonography/Ultrasound Imaging
In this technique, a high-frequency sound wave is used to generate an image. Utrasonography is made use of in heart related organs and in the abdomen for the evaluation of a fetus (Popov et. al., 2002).
Radioisotope imaging
This method uses radioactive emissions for definition of a condition. On the general note, these tools have become the key drivers of medicine and are effective in predicting, diagnosing, as well as in treating malfunction of body parts in patients (Hasuike et. al., 2002).
Heterogeneity in DCE-MRI
The heterogeneity of tumors is mainly described by the various rates of perfusion (Gaustad et. al, 2005). Most researchers have analyzed heterogeneity through the use of quantitative methods of assessing volumetric changes in specific tumoral areas. A study by Kemeny and Fata (1999) revealed that an evaluation of tumor heterogeneity is fundamental in the description of treatment methods and objectives. The researchers in their paper emphasized the utilization of machine learning approaches in the production of automatic and usable segmentation findings in the quantitative analysis of DCE-MRI data sets (Kemeny and Fata, 2001). Daducci et al., (2001) applied machine learning technique in the analysis of DCE-MRI to assess the effect of treatment with an experimental anticancer therapy. In their study, the SVM had been trained to detect tumor regions of contrast agent accumulation and regions with negligible enhancement originating from necrotic tissue (Daducci et al., 2001).
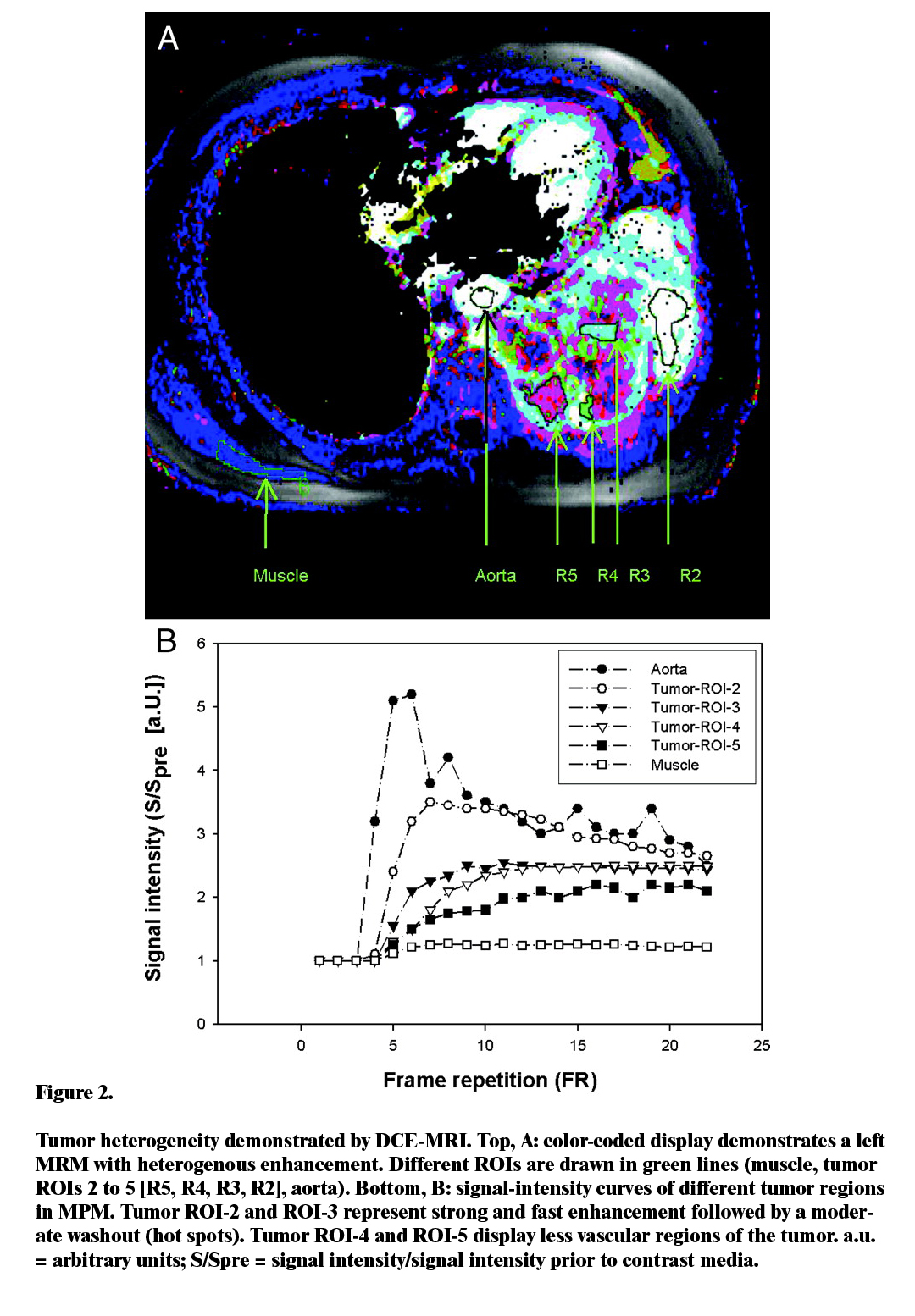
Similar studies have revealed that there is an appreciable rise of the level of necrotic volumes on patients that were not offered any treatment, hence illustrating the importance of heterogenicity distinguishing when using DCE-MRI (Figure 2).
For this study, two critical values of tumour vascular heterogeneity were discovered to be statistically significant. These measures are the ratio enhancing tumour voxels to total tumor voxels, and the box dimension. The former measure was particularly sensitive to the treatment effect associated with anti-angiogenic and anti-vascular elements. High figures in the ratio were found to indicate that the tumor was well perfused and that medications administered though systemic routes could easily go through the walls of the tumor. As such, the analysis found it safe to conclude that the ratio is a non-specific marker whose main influences include physiological aspects such as the permeability and interstitial pressure. On the other hand, it is also important to consider the fact that linkage between various biomarkers and a change in the size of the tumor is heavily dependent on the type of therapy used, as well as the inherent characteristics of the tumor.
A research conducted by Goldberg (2001) confirmed that spatial heterogeneity statistics can provide data that is naturally not accessible using distribution-based techniques. They also illustrated that the extent of heterogeneity increases with the complexity of the item being assessed. Further findings from the experiment include the fact that spatial heterogeneity statistics respond according to the change that may present in clinical experiments. A number of factors could affect the findings of a spatial heterogeneity evaluation. As such, researchers should take absolute care when implementing proposed statistics that are presented as adjuncts to traditional methodologies. Spatial heterogeneity evaluation methods use a combination of data on the shape, size and constituency of the tumor as well as the DCE-MRI settings. However, it is worth noting that a number of these factors can be modified hence affecting the eventual interpretation of findings. For instance, an individual tumor could contain cystic components that change in the duration of the study hence affecting the shape and size of such a swelling. Consequently, a spatial analysis conducted on a tumor that has the undergone such changes would definitely not present a clear picture. Aside from this, movement of the tumor while conducting the assessment would eventually lead to poor parameter approximation and a wrong evaluation of heterogeneity. It is for this reason that DCE-MRI analyses are required to include extensive quality checks.
Predicting tumour shrinkage in bevacizumab plus FOLFOX-6 Dataset
Following the establishment of the importance of heterogeneity, O’Connor et al. (2011) conducted further tests to establish the effect of introducing a combination of Bevacizumab and FOLFOX-6 to the cancerous tissues. They concluded that CRC liver shrinkage due to Bevacizumab and FOLFOX-6 is predictable by DCE-MRI biomarkers of tumour heterogeneity (O’Connor et al., 2011).
For this research, an investigation of the relative value of a number of pre-treatment biomarkers of tumor blood supply has been conducted. In addition, the study has also tried to evaluate the reduction of tumor size brought about by bevacizumab and FOLFOX-6 therapy. My guiding hypothesis was that the size reduction of tumours can be evaluated using baseline image information. However, it has been discovered that basic measure of size cannot be used independently in the prediction of clinical outcome. For the current study, over 80% of the percentage change in the size of the tumor can be described by assessing a combination of imaging biomarkers. Results were given validation by another set of pre-treatment information collected from the same growth. From the study, it was discovered that the size of the tumor can be statistically important in the provision of complementary values of data about the location of the tumor. These findings support those of other studies that have presented image analysis from various parameters as for prediction of clinical outcome.
The extended Tofts model and DCE-MRI
While using DCE-MR Imaging, it is mandatory that the perfusion of tissues and blood volume be well estimated if proper treatment is to be prescribed. The Extended Tofts Model has been regarded as the best way to quantify tumor vascularity (Liu et. al., 1999). This model works by repeatedly imaging of tissues by use of a T1 weighted sequence to estimate the amount of time it takes for a contrast agent to cross through blood barriers. A specific mathematical compartmental structure is then used to provide approximations of the permeability of tissues. It is, however, worth noting that the Extended Tofts model is very accurate in tissues that have very little vasculature (O’Connor et al., 2011). A combination of the values obtained with estimates of blood volume helps accord appropriate treatment.
Advantages of MRI
A particular merit that is known of the MR liver-imaging is the fact that is effects the softening of tissue contrasts thereby revealing more acceptable characterization of focal-liver-lesions which may be considered. Studies have shown that, “The development of a liver-specific MR contrast agent has further improved the diagnostic yield of MRI in lesion detection and characterization” (Abramson et. al., 2000).
The CT/MR imaging context of liver-metastases is identified. For a more defined prediction from the use of this medical imaging device, there is emphasis on the key-role which the MRI presents when compared with CT for assessing the condition of patients with the extraheptic-cancer condition as well as limited-liver-metastases-this is considered when the patients are candidates for surgical operations.
Studies conducted by Li et al,(1999) reviewed usage of Enhanced Contrast US (CEUS) for improving the sensitiveness or the US specific usage through differentiating malignant-vs-benign at about 78%-100% from 23%-92%, respectively. Equally, Okuno et al., (1999) studied diagnostic-yield of CEUS and helical-CT for predicting liver-metastases and noted a CEUS of 97% when lesions was observed using CT. In any case, the studies did not reveal any histological-standards for referencing availably determined sensitivity. Presently, CUES presents a clearly defined role in evaluating and predicting equivocal-lesions in the US-convention and in the monitor of reaction to treatment after there has been a localized tumor-therapy. But based on the shortcomings witnessed in visualizing sectional distributions of 3D-shaped metastases, there has been the limitation in pre-operating the patient assessment using colorectal-liver-metastases. In any case, contrasting agents have brought about an increment in diagnostic-yield of intraoperative-US which impacts on strategic surgery.
Imaging-Based Clinical Trials
Clinical trials have a very fundamental placement in testing a newly produced drug or a device in present-day medical practices. Medical-imaging recently has assumed so much significance in clinical-trials due to the fact that images make available highly precise pictorial definition of a diagnosed condition in quantifiable parameters. According to studies, a basic imaging-based clinical trial is made up of the following three things:
- A well-defined rigorous clinical trial protocol (Gillams and Lees, 2000).
- A radiology core that has a quality control mechanism, a biostatistics component, and a server for storing and distributing data and analysis results (Gillams and Lees, 2000). 3) Many field sites that generate and send image studies to the radiology core (Gillams and Lees, 2000).
With the general increment witnessed in clinical trials, there is an equally associated difficulty with the service of fundamentals of radiology in serving adequately as administrative or distributive mechanism for participatory clinician/radiologists at the global screen. From studies, it has been gathered that as more and more clinical trial develop, the biggest issue facing practitioners in the radiology is to develop proper servers that can comfortably handle multiple trials. This is in addition to the meeting of requirements for a quick distribution of data to other radiologists (Dancey et. al., 2000).. According to Dancey et al, “Data Grid in the grid computing technology can satisfy the aforementioned requirements of imaging-based clinical trials “(2000).
The CRC presents a clearly defined role in evaluating and predicting equivocal-lesions in the US-convention and in the monitor of reaction to treatment after there has been a localized tumor-therapy. But based on the shortcomings witnessed in visualizing sectional distributions of 3D-shaped metastases, there has been the limitation in pre-operating the patient assessment using colorectal-liver-metastases.
For a more defined prediction from the use of medical imaging devices, there is emphasis on the key-role which the MRI presents when compared with CT for assessing the condition of patients with the extra-hepatic cancer condition as well as limited-liver-metastases-this is considered when the patients are candidates for surgical operations.
Methodology
The Sampling Method
The evaluator will draw a total of ten patients with 26 tumors in the oncology unit for assessment. An opportunistic sampling was chosen for this project because it is the least time consuming method of obtaining data from a given group of people. The sample is predefined thus eliminates possibilities of sampling bias. It also costs less to do an opportunistic sampling and despite this, the results obtained are usually an accurate reflection of the situation in the population. All patient records will be extracted from the center database, checked to see which patient and treatment prediction method classification they belong to and listed as part of the project sampling frame. A sample frame of 10 was chosen as an ideal figure for research because it would provide for effective data collection at an effective cost.
Ethical Considerations
Approval for this evaluation project was obtained from the University Ethics Committee. Consequently, the evaluator was commited to suppress any information that might lead to unmistakably identifying individual participants. Informed consent from the patients was obtained and for legal purposes this included the signing of confidentiality agreement.
Regression
Various statistical methods of presenting the relationship between a number of factors have been developed. Regression is a statistical tool that allows the investigator to study the relationship between two variables. Basically it helps in examining the causal effect of one variable on another. For instance in the case of cancer, tumor growth can be said to be linked to the development of new vasculature and the number muscles around the tumor among other things. In order to clearly show how the two latter elements relate with the former (tumor growth), a statistical analysis has to be conducted and the findings plotted on a curve that presents this. There are two types of regression, i.e., linear regression and non-linear regression. Using the aforementioned example, if all the other causative elements of cancer are frozen, and only the impact of one (say the extent of vasculature) is found to determine the growth of the lesion, then a linear relation is seen to have occurred. On the other hand, if a number of elements that interrelate with each other are used for assessing the consequence of another variable, then a multivariate or non-linear regression is used to present the findings. The section below describes these basic regression models in view of their relevance to the current project.
Linear regression
Linear regression deals with causative relationship between two variables. For instance cancer has many causative elements. Should the investigator decide to freeze all the other elements and only consider the impact of one (say the extent of vasculature) on the growth of the lesion, then the relationship becomes linear and hence linear regression. It is the responsibility of radiologic-imaging to analyze the condition of the liver by assessing the availability or the non-availability of liver-metastases when considering surgery of better predictions as well as for evaluating successes of other patients’ chemotherapy even though there has been the use of transabdominal-sonography in assessing the liver. The method is not without its shortcomings: it ignores other causative elements which are equally important in understanding the case. In the illustration above, there has to be a consideration of the operator’s level of understanding which in most cases reflects the reviewer of equivocal-results in patients who (induced with chemotherapy) express fatty liver infiltrations (Copur et. al., 2001).
Non-linear modeling
Unlike linear regression, non-linear regression examines the causative relationship between many variables. It considers all the elements that interrelate with one another when examining the consequence of another variable. It main strength lies in the fact that it ensures that all causative elements are considered when examining a problem. This study used a non-linear model of regression analysis. Out of the 26 patients selected, the best approach would be to pick ten to guide the study as this was seen to be ideal for the best levels of accuracy.
Ensemble learning-Boosting
Boosting is arguably the most reliable and most effective of all ensemble learning ensembles (Cromheecke et. al., 1999; Copur et. al., 2001). Boosting works by incrementally developing sets of alternative model through provision of guidance mechanisms for directing individual model instances to enhance the training instances, which other previously developed earlier, failed to analyze properly. The main reason as to why boosting works properly given any set of data is that it emphasizes accuracy through the repetitive enhancement of training instances. Adaboost is the most commonly used algorithm when it comes to imaging using both linear and nonlinear regression methods.
Bootstrap aggregating
This is an Ensemble Learning approach that works by ensuring that individual models in the statistical group compete equally. The strategy, commonly referred to as bagging, guides each model in the ensemble by use of a randomly-developed underling of the training group. To achieve the highest classification accuracy, especially when using random regression models, a combination of randomly selected trees (the random forest algorithm) with bootstrap aggregating works best (Cromheecke et. al., 1999; Copur et. al., 2001).
Errors in variable regression
Every research has to deal with errors in the data. At times the errors act as a positive step upon which the investigator can further model the data. For this particular research, the stepwise errors-in-variable regression helped model the reduction of the tumor by use of the statistics availed before treatment. The CT/MR imaging context of liver-metastases was identified. For a more defined prediction from the use of this medical imaging device, there was emphasis on the key-role which the MRI presents when compared with CT for assessing the condition of patients with the extraheptic-cancer condition as well as limited-liver-metastases. This was considered with the patients who were candidates for surgical operations (van Riel et. al., 2000). This provided an opportunity for linear regression. The regression was redone using data from a repeat scan to offer validation to the results.
It is the responsibility of radiologic-imaging to analyze the condition of the liver by assessing the availability or the non-availability of liver-metastases when considering surgery of better predictions as well as for evaluating successes of other patients’ chemotherapy even though there has been the use of transabdominal-sonography in assessing the liver, the method is not without its shortcomings: there has to be a consideration of the operator’s level of understanding which in most cases reflects the reviewer of equivocal-results in patients who (induced with chemotherapy) express fatty liver infiltrations (Cromheecke et. al., 1999; Copur et. al., 2001).
How missing data was handled
The missing data for a number of patient’s initial prediction of treatment scans were accordingly dealt with by deleting and imputation from the various generated lists. This was done in order to ensure that the amount of data present was used to the maximum, while at the same time reducing levels of bias.
Re-sampling
Re-sampling is usually important for validation purposes. In studies of this nature, re-sampling refers to a statistical model involved in the modification of the dimensions of images developed using digital techniques through re-analyzing the original set of information. Various sample points are identified depending on the distance from the original center and are assigned special identification color values. This technique involves multivariate interpolation where the closest centers to particular sample points are selected and analyzed irrespective of the influences of other surrounding centers.
Conclusion
Knowledge of the extent of a tumor is fundamental in the staging of the solid tumor and as such important in the selection of treatment methods as well as the assessment of the applicability of resection in addition to the prediction of clinical results. However, the connection between the size of the tumor before treatment and the clinical outcome is not always linear and therefore not reliable as a statistical relationship. Usable data can help in the prediction of treatment outcomes though the usage of techniques such as Dynamic Contrast Enhanced MRI (DCE-MRI) which offer more opportunities for analyzing a tumour’s pathophysiology. For example, basic assessment values such as high baseline DCE-MRI before the administration of treatment have presented a statistically significant link with the beneficial treatment outcome succeeding a number of cytotoxic medications in persons with CRC (Stubbs et al, 2001). Biomarkers used in the prediction of treatment regimens are very important in the development of individualized medicine. Various researches have made efforts to analyze predictive biomarkers depending on the size of the tumour before treatment but most have not been very successful. The current study has illustrated that the provision of data describing the micro-vascular structure by using Magnetic Resonance Imaging (MRI) can substantially help in proper treatment prediction.
The study was however limited in some form. The reliability of the evaluation results apply solely to the cohort of patients who had undergone treatment prediction at a number of facilities. It would take a great deal of resources and time to draw a nationally representative sample of patients. Due to time constraint, the evaluator settled on a small sample frame of only ten patients. The results of this study may thus not be representative enough.
Despite its limitations, this investigation has provided primary evidence that the concurrent usage of various pre-treatment measures obtained from DCE-MRI parameter maps may be used reliably in the predication of tumor shrinkage. This is in response to the clinical low-error combination of bevacizumab and cytotoxic chemotherapy. The evaluation has hypothesized that the model can be described by assessing the penetration of the medication and its accumulation. The uniformity of tumor vasculature can also help describe the model. A combination of biomarkers is valuable in the identification of customized tumor treatment therapies. Such results create more room for the assessment of image heterogeneity in oncology studies using Magnetic Resonance imaging.
Reference List
ACCP ,2011. Dynamic Contrast-Enhanced MRI of Malignant Pleural Mesothelioma. USA: American College of Chest Physicians
Abramson, R. Rosen, M. Perry, J. Brophy, P. Raeburn, L. and Stuart, K., 2000. Cost- Effectiveness of Hepatic Arterial Chemoembolization for Colorectal Liver Metastases Refractory to Systemic Chemotherapy. Radiology, 4(216), pp.485- 491.
Bloom, A.I. Gordon, R.L. Ahl, K.H. Kerlan, R.K. LaBerge, J.M. Wilson, M.W. Venook, A.P. and Warren, R., 1999. Transcatheter embolization for the treatment of misperfusion after hepatic artery chemoinfusion pump implantation. Ann Surg Oncol, 3(2), pp.350-358.
Buchegger, F. Allal, A.S. Roth, A. Papazyan, J.P. Dupertuis, Y. Mirimanoff, R.O. Gillet, M. Pelegrin, A. Mach, J.P. and Slosman, D.O., 2000. Combined radioimmunotherapy and radiotherapy of liver metastases from colorectal cancer: a feasibility study. Anticancer Res, 4(20), pp.1889-1196.
Choi, H. Loyer, E.M. DuBrow, R.A. Kaur, H. David, C.L. Huang, S. Curley, S. and Charnsangavej, C., 2001. Radio-frequency ablation of liver tumors: assessment of therapeutic response and complications. Radiographics, 2(21), S41-54.
Copur, M.S. Capadano, M. Lynch, J. Goertzen, T. McCowan, T. Brand, R. and Tempero, M., 2001. Alternating hepatic arterial infusion and systemic chemotherapy for liver metastases from colorectal cancer: a phase II trial using intermittent percutaneous hepatic arterial access. J Clin Oncol, 6(19), pp.2404- 2412.
Cromheecke, M. de-Jong, K.P. and Hoekstra, H.J. 1999. Current treatment for colorectal cancer metastatic to the liver. Eur J Surg Oncol, 9(25), pp.451-463
Curley, S.A. Izzo, F. Delrio, P. Ellis, L.M. Granchi, J. Vallone, P. Fiore, F. Pignata, S. Daniele, B. and Cremona, F., 1999. Radiofrequency ablation of unresectable primary and metastatic hepatic malignancies: results in 123 patients. Ann Surg 17(230) 1-8
Dancey, J.E. Shepherd, A. Paul, K. Sniderman, K.W. Houle, S. Gabrys, J. Hendler, A.L. and Goin, J.E., 2000. Treatment of nonresectable hepatocellular carcinoma with intrahepatic 90Y-microspheres. J Nucl Med, 9(41), pp.1673-1681.
Fiorentini, G. Poddie, D.B. Giorgi, U.D. Guglielminetti, D. Giovanis, P. Leoni, M. Latino, W. Dazzi, C. Cariello, A. Turci, D. and Marangolo, M., 2000. Global approach to hepatic metastases from colorectal cancer: indication and outcome of intra- arterial chemotherapy and other hepatic-directed treatments. Med Oncol, 2(17), pp.163-173
Gaustad, J., Benjaminsen, I.C., Graff, B.A. et. al., 2005. Intratumor heterogeneity in blood perfusion in orthotopic human melanoma xenografts assessed by dynamic contrast-enhanced magnetic resonance imaging. Journnal of Magnetic Resonance Imaging, 21(6), pp. 792-800.
Geoghegan, J.G. and Scheele, J., 1999. Treatment of colorectal liver metastases. Br J Surg, 29(6) pp.158-169.
George M.L., Dzik-Jurasz A.S., Padhani AR, et al., 2001. Non-invasive methods of assessing angiogenesis and their value in predicting response to treatment in colorectal cancer. Br J Surg, 88, pp. 1628-1636.
Gillams, A.R. and Lees, W.R., 2000. Survival after percutaneous, image-guided, thermal ablation of hepatic metastases from colorectal cancer. Dis Colon Rectum, 12(43), pp. 656-661.
Glover, C. Douse, P. Kane, P. Karani, J. Meire, H. Mohammadtaghi, S. and Allen- Mersh, T.G., 2000. Accuracy of investigations for asymptomatic colorectal liver metastases. Dis Colon Rectum, 12(45), pp. 476-484.
Goldberg, S.N., 2001. Radiofrequency tumor ablation: Principles and techniques. Eur J Ultrasound, 2(13), pp. 129-147.
Hasuike, Y. Takeda, Y. Mishima, H. Nishishou, I. Tsujinaka, T. and Kikkawa, N., 2002. Systemic chemotherapy for advanced colorectal cancer with liver metastasis. Gan To Kagaku Ryoho, 20(29), pp.866-872.
Heslin, M.J. Medina-Franco, H. Parker, M. Vickers, S.M. Aldrete, J. and Urist, M.M., 2001. Colorectal hepatic metastases: resection, local ablation, and hepatic artery infusion pump are associated with prolonged survival. Arch Surg, 28(136), pp.318-323.
Johns LE, Houlston RS., 2001.A systematic review and meta-analysis of familial colorectal cancer risk. Am J Gastroenterol, 23(96), pp. 2992-3003.
Kemeny, N. and Fata, F., 2001. Hepatic-arterial chemotherapy. Lancet Oncol, 2(2), pp.418-428
Landheer, M.L. Therasse, P. and van de Velde, C.J., 2001. The importance of quality assurance in surgical oncology in the treatment of colorectal cancer. Surg Oncol Clin N Am, 2(10), pp.885-914.
Lode, H.N. Xiang, R. Becker, J.C. Gillies, S.D. Reisfeld, R.A., 1998. Immunocytokines: a promising approach to cancerimmunotherapy. Pharmacol Ther, 19(80), pp. 277-292.
Li, L. Wu, P.H. Mo, Y.X. Lin, H.G. Zheng, L. Li, J.Q. Lu, L.X. Ruan, C.M. and Chen, L., 1999. CT arterial portography and CT hepatic arteriography in detection of micro liver cancer. World J Gastroenterol, 9(5), pp.225-227.
Link, K.H. Sunelaitis, E. Kornmann, M. Schatz, M. Gansauge, F. Leder, G. Formentini, A. Staib, L. Pillasch, J. and Beger, H.G., 2001.Regional chemotherapy of nonresectable colorectal liver metastases with mitoxantrone, 5-fluorouracil, folinic acid, and mitomycin C may prolong survival. Cancer, 20(92), pp.2746-2753.
Liu, L. Jiang, Z. Teng, G.J. Song, J.Z. Zhang, D.S. Guo, Q.M. Fang, W. He, S.C. and Guo, J.H., 1999. Clinical and experimental study on regional administration of phosphorus 32 glass microspheres in treating hepatic carcinoma. World J Gastroenterol, 9(5), pp. 492-505.
Lorenz, M. Staib-Sebler, E. Hochmuth, K. Heinrich, S. Gog, C. Vetter, G. Encke, A. and Muller, H.H., 2000. Surgical resection of liver metastases of colorectal carcinoma: short and long-term results. Semin Oncol, 12(27), pp.112-119.
Meyer C.R., Armato S.G., Fenimore CP, et al., 2009. Quantitative imaging to assess tumor response to therapy: common themes of measurement, truth data, and error sources. Transl Oncol, 2, pp.198-210.
McNulty, C., 2011. Rotator Cuff MRI Shoulder Scan & Arthrogram (MRA). Web.
Neeleman, N. Wobbes, T. Jager, G.J. and Ruers, T.J., 2001. Cryosurgery as treatment modality for colorectal liver metastases.Hepatogastroenterology, 20(48), pp.325- 359.
O’Connor, J.P.B., Rose, C.J., Jackson, A. et al., 2011. DCE-MRI biomarkers of microvascular structure and function predict CRC liver metastasis shrinkage induced by bevacizumab and FOLFOX-6. British Journal of Cancer 105, 139-145
Okuno, K. Kaneda, K. and Yasutomi, M., 1999. Regional IL-2-based immunochemotherapy of colorectal liver metastases. Hepatogastroenterology, 19(46), pp.1263-1267.
Popov, I. Lavrnic, S. Jelic, S. Jezdic, S. and Jasovic, A., 2002. Chemoembolization for liver metastases from colorectal carcinoma: risk or a benefit. Neoplasma, 23(49), pp.43-48
Primrose, J.N., 2002. Treatment of colorectal metastases: surgery, cryotherapy, or radiofrequency ablation. Gut, 15(50), pp. 1-5.
Ragnhammar, P. Hafstrom, L. Nygren, P. and Glimelius, B., 2001.SBU-group: Swedish Council of Technology Assessment in Health Care. A systematic overview of chemotherapy effects in colorectal cancer. Acta Oncol, 20(40) pp.282-308.
Ruers, T.J. Joosten, J. Jager, G.J. and Wobbes, T., 2001. Long-term results of treating hepatic colorectal metastases with cryosurgery. Br J Surg 26(88), pp.844-849.
Stubbs, R.S. Cannan, R.J. and Mitchell, A.W., 2001.Selective internal radiation therapy (SIRT) with 90Yttrium microspheres for extensive colorectal liver metastases. Hepatogastroenterology, 16(48), pp.333-337.
van Riel, J.M. van Groeningen, C.J. Albers, S.H. Cazemier, M. Meijer, S. Bleichrodt, R. van den Berg, F.G. Pinedo HM, and Giaccone, G., 2000. Hepatic arterial 5- fluorouracil in patients with liver metastases of colorectal cancer: single-centre experience in 145 patients. Ann Oncol, 2(11), pp.1563-1570.
Zealley, I.A. Skehan, S.J. Rawlinson, J. Coates, G. Nahmias, C. and Somers, S., 2001.Selection of patients for resection of hepatic metastases: improved detection of extrahepatic disease with FDG pet. Radiographics, 2(21) S55-69